

- [email protected]
+91 40 2344 5962 - View Stock Quote


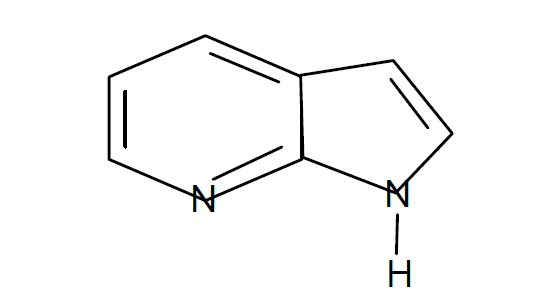

Synonyms : 1H – Pyrrolo[2,3-b]pyridine
CAS No. : [271-63-6]
Molecular Formula : C7H6N2
Formula weight : 118.14
7-Azaindole is a heterocyclic molecule tht can be utilized as a pharmaceutical building block, starting material in a recent synthesis of azaserotonin.
Azaindoles have gained sustainable synthetic interest primarily as bioisostere for indoles. The naturally occurring 7-azaindoles are relatively scarce compared to indoles. In its replacement, even Variolins are used which were generally isolated from Antarctic sponge Kirkpatrick Variolosa. Variolin B is considered as the most active derivative among all these alkaloids against P388 murine leukemia cells.
7- Azaindoles have been an essential target of extensive synthetic efforts as a consequence of their potent pharmacological activities. The most basic method for the construction of the ring system involves pyrrolo annelation into a preformed pyridine ring. The other available routes to 7- Azaindoles such as Madelung [17, 18] or Fischer [19, 20] are often ignored or limited mainly due to their narrow scope compared to their use in indole field.
The few properties of some 7 - Azaindoles with respect to plants have been studied by various scientists. It is witnessed that the vacuolation of protoplasms in the roots of Allium cepa is stimulated by the 7 -azaindoles-3-acetic acid.
This compound has also been the subject of intense investigation by fluorescence spectroscopy. Also, at room temperature, the proton transfer reaction leads to tautomers at a fast rate with essentially no dimeric emissions.
It is used as a heterocyclic molecule that finds its application in the pharmaceutical building blocks. It forms the starting material in a new synthesis of azaserotonin. The 7- azatryptophan inhibits the growth of Nicotiana tabacum and Daucus carota cells. 7-azaindole derivatives have been effectively tested as potential agrochemicals. Many Biological activities were also observed with enzymes or bacteria.
7-azaindole tends to bind to the enzyme Erm (methyltransferase family) which in turn confers resistance to antibiotics. It is also believed to decrease the Tetrahymena pyriformis growth. They have widespread application as potential pharmaceutical agents.